Many patients assume all medical treatments work the same way for everyone. This outdated belief ignores how our unique genetic profiles, environments, and lifestyles influence treatment effectiveness. Personalized healthcare offers a smarter approach by matching therapies to your individual characteristics, potentially improving outcomes and reducing adverse reactions. This guide explains what personalized healthcare means, how it works through advanced testing methods, where it’s currently applied with proven results, and practical steps you can take to access these tailored treatment options for yourself or your family.
Table of Contents
-
Applications And Evidence Of Benefits In Personalized Healthcare
-
Nuances, Challenges, And Differences: Personalized Vs Precision Medicine
-
How Patients And Families Can Apply Personalized Healthcare Today
Key takeaways
| Point | Details |
|---|---|
| Personalized healthcare definition | Medical treatment tailored to individual genetics, environment, and lifestyle rather than standardized protocols. |
| Core methodologies | Uses genomic sequencing, pharmacogenomics, biomarker identification, and AI analysis to customize care. |
| Proven applications | Precision oncology and pharmacogenomics show improved response rates and progression-free survival in trials. |
| Important distinctions | Personalized medicine is holistic including lifestyle; precision medicine focuses on genomics and data-driven approaches. |
| Practical considerations | Costs vary widely, insurance coverage is inconsistent, and patients should discuss testing benefits with providers. |
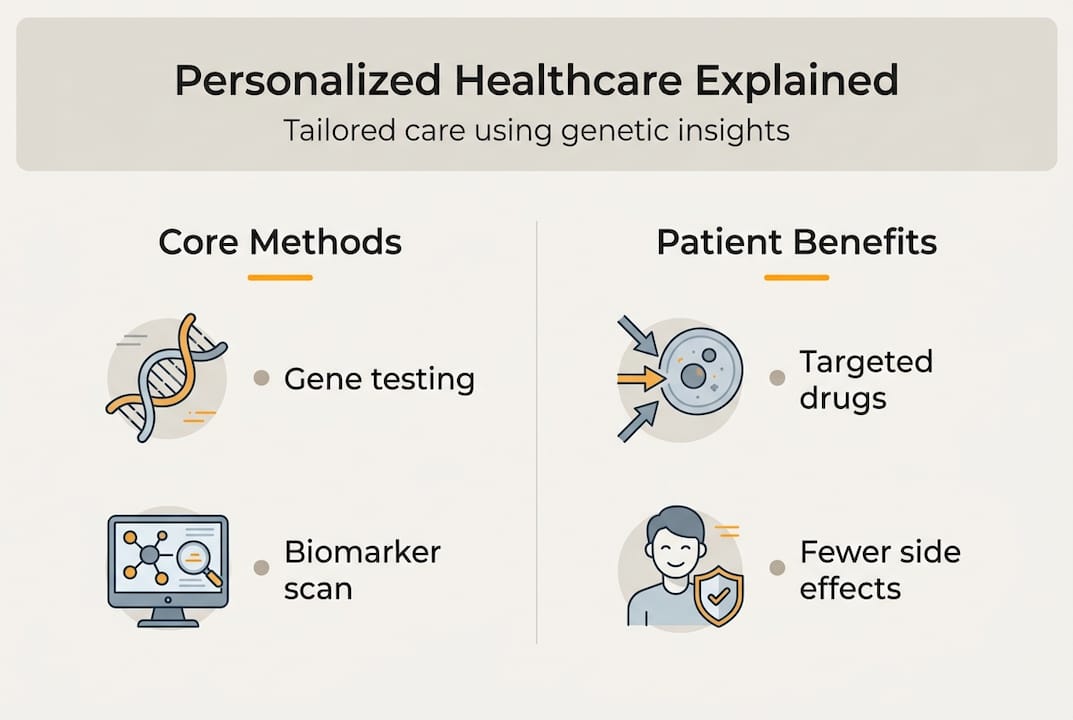
What is personalized healthcare and how does it work?
Personalized healthcare tailors medical treatment to individual patients based on their genetic makeup, environment, lifestyle, and other factors, moving away from a one-size-fits-all approach. Traditional medicine often prescribes the same drug at the same dose to all patients with a given condition. Personalized healthcare recognizes that two people with identical diagnoses may respond completely differently to the same treatment because of their unique biological profiles.
Several key factors shape personalized treatment plans. Your genetic variants influence how you metabolize medications, whether you’re predisposed to certain diseases, and which therapies will work best for your body. Environmental exposures like pollution, diet, and occupational hazards also play crucial roles. Lifestyle choices including exercise habits, stress levels, and sleep patterns round out the picture your healthcare team considers when designing your treatment.
Core methodologies make personalization possible. Next-generation sequencing (NGS) rapidly analyzes your entire genome or specific genes to identify mutations that affect disease risk or drug response. Pharmacogenomics studies how your genes influence medication effectiveness and side effects, allowing doctors to prescribe the right drug at the right dose from the start. Biomarker identification finds measurable indicators in your blood, tissue, or other samples that predict treatment response. AI analysis processes massive datasets to spot patterns humans might miss, connecting your profile to optimal treatment strategies.
These technologies work together at a medical center equipped for personalized care. A simple blood draw or cheek swab provides the genetic material needed for analysis. Laboratory specialists sequence your DNA and identify relevant variants. Bioinformatics tools compare your results against vast databases of genetic information and treatment outcomes. Your physician interprets these findings alongside your medical history, current symptoms, and personal preferences to craft a treatment plan specifically for you.
Understanding precision medicine helps you have informed conversations with your healthcare team. The field evolves rapidly as researchers discover new genetic markers and treatment connections. What seemed impossible five years ago is now routine practice in many specialties.
Pro Tip: Ask your provider which genetic tests are available for your condition and how results might change your treatment plan before committing to testing.
Applications and evidence of benefits in personalized healthcare
Personalized healthcare shows the strongest evidence in oncology, where tumor profiling guides targeted therapy selection. Doctors analyze cancer cells to identify specific mutations driving tumor growth. Targeted drugs then attack those exact vulnerabilities while sparing healthy tissue. This approach produces better outcomes than traditional chemotherapy for many cancer types.

Applications include precision oncology, pharmacogenomics for dosing, rare diseases, and chronic conditions like cardiovascular disease. In precision oncology, next-generation sequencing identifies actionable mutations in genes like EGFR, ALK, or BRAF. Patients with these mutations receive drugs specifically designed to block the mutated proteins. Pharmacogenomics customizes medication dosing based on genetic variants affecting drug metabolism. The gene CYP2C9, for example, influences how you process warfarin, a common blood thinner. Testing for CYP2C9 variants helps doctors prescribe the safest, most effective warfarin dose for your body.
Clinical trial data demonstrates real benefits. The ROME trial evaluated tumor-informed therapy across multiple cancer types and showed improved response rates compared to standard treatment. Patients whose therapies matched their tumor’s molecular profile experienced longer progression-free survival. The TuPro melanoma study found similar advantages when treatments aligned with genetic testing results.
| Study | Cancer Type | Personalized Approach | Standard Care Response | Personalized Response | Benefit |
|---|---|---|---|---|---|
| ROME | Multiple | Tumor profiling | 28% | 41% | 13% improvement |
| TuPro | Melanoma | Genetic matching | 35% | 52% | 17% improvement |
| Lung Cancer | NSCLC | EGFR targeting | 10 months PFS | 18 months PFS | 8 months longer |
Chronic disease management also benefits from personalization. Cardiovascular patients with specific genetic variants may need different statin doses or alternative cholesterol medications. Diabetes treatment increasingly incorporates genetic testing to predict which patients will respond to metformin versus other glucose-lowering drugs. Psychiatric care uses pharmacogenomic panels to identify antidepressants and antipsychotics most likely to work without causing intolerable side effects.
The medical center approach integrates these testing capabilities into routine care pathways. Patients receive genetic counseling before testing to understand potential results and implications. Laboratory teams process samples using validated protocols. Physicians trained in genomic medicine interpret results and translate them into actionable treatment modifications.
Pro Tip: Ask your healthcare team about published clinical trial data specific to your condition and how personalized approaches might improve your individual outcomes.
Nuances, challenges, and differences: personalized vs precision medicine
The terms personalized medicine and precision medicine are often used interchangeably, but important distinctions exist. Understanding these differences helps you navigate conversations with healthcare providers and set realistic expectations.
| Aspect | Personalized Medicine | Precision Medicine |
|---|---|---|
| Definition | Holistic approach including genetics, lifestyle, preferences, environment | Data-driven approach focused primarily on genomics and molecular profiling |
| Scope | Broader, encompasses all individual factors | Narrower, emphasizes measurable biological data |
| Example | Treatment plan considers your work schedule, family support, dietary preferences alongside genetics | Treatment selected based solely on tumor mutation profile |
| Tools | Genetic tests plus lifestyle assessments, patient interviews, preference surveys | Next-generation sequencing, biomarker panels, AI algorithms |
Personalized healthcare includes lifestyle and preferences making it holistic; precision medicine is data-driven via genomics and AI. Limitations include costs, access disparities, data bias, and privacy concerns. Both approaches face significant challenges that affect patient access and outcomes.
Cost represents the most immediate barrier. Comprehensive genomic sequencing ranges from $1,000 to $5,000 depending on the panel’s breadth. Tumor profiling for cancer patients can exceed $10,000. Many insurance plans cover testing only for specific conditions or after standard treatments fail. Patients in rural areas or developing regions often lack access to facilities offering advanced testing and interpretation services.
Data bias poses a scientific challenge. Most genomic databases contain predominantly European ancestry samples. This means genetic variants common in Asian, African, or Latin American populations may be misclassified or poorly understood. Treatment recommendations based on biased data could be less effective or even harmful for underrepresented groups. Researchers are working to diversify genomic databases, but progress remains slow.
Privacy concerns worry many patients considering genetic testing. Your DNA reveals information not just about you but also about your relatives. AI challenges in personalized medicine include the black box problem where algorithms make recommendations without transparent reasoning. Data breaches could expose sensitive health information to employers, insurers, or other parties who might discriminate based on genetic risk factors. Strong legal protections exist in some countries, but enforcement varies.
Ethical challenges extend beyond privacy. Should parents test children for adult-onset disease risks? How do we ensure equitable access when personalized treatments cost significantly more than standard care? Who owns your genetic data once it’s sequenced? These questions lack universal answers and require ongoing dialogue between patients, providers, ethicists, and policymakers.
Clinician oversight remains essential despite technological advances. AI tools assist with data analysis but cannot replace human judgment. Your doctor considers test results alongside physical examination findings, medical history, and your personal values when recommending treatment. Clinical trial evidence should guide decisions whenever possible rather than relying solely on theoretical predictions from genetic data.
Practical considerations for patients include asking about total costs upfront, understanding what insurance will cover, questioning how test results will change your treatment, learning how your data will be stored and who can access it, and requesting second opinions when recommendations seem unclear or overly aggressive.
The medical center environment should provide transparent information about costs, data handling, and the evidence base supporting personalized recommendations.
How patients and families can apply personalized healthcare today
You don’t need to wait for your doctor to suggest personalized healthcare. Proactive patients can initiate conversations and explore options that might improve their treatment outcomes. Follow these practical steps to incorporate personalized approaches into your healthcare journey.
-
Learn about personalized options relevant to your condition by researching which genetic tests or biomarker panels apply to your diagnosis. Cancer patients should ask about tumor profiling. People starting new medications can inquire about pharmacogenomic testing to predict drug response. Patients with strong family histories of disease may benefit from genetic risk assessments.
-
Ask providers about testing availability and whether results would change your treatment plan. Not every condition benefits equally from personalized approaches. Your doctor should explain which tests are evidence-based for your situation versus experimental. Request information about test accuracy, turnaround time, and how results will be interpreted.
-
Understand insurance coverage and out-of-pocket costs before proceeding. Contact your insurance company directly to verify coverage for specific tests. Many genetic testing companies offer financial assistance programs for patients who qualify. Compare costs across different laboratories as prices vary significantly for identical tests.
-
Evaluate benefits versus limitations honestly. Personalized healthcare can improve outcomes by matching treatments to your unique profile but not all cases benefit equally; discuss costs and access with your provider. Some genetic findings have unclear significance, leading to anxiety without actionable treatment changes. Consider whether you’re prepared to handle uncertain results.
-
Stay informed on ongoing research and new testing options. The personalized medicine field evolves rapidly with new discoveries published monthly. Ask your healthcare team about clinical trials investigating personalized approaches for your condition. Patient advocacy groups often share updates about emerging testing technologies and treatment options.
Realistic expectations matter enormously. Genetic testing won’t provide all answers and some results may be inconclusive. Personalized treatments aren’t guaranteed to work better than standard options for every patient. The process takes time as samples must be collected, analyzed, and interpreted before treatment modifications occur.
Holistic care extends beyond genetic testing. Your lifestyle choices profoundly impact treatment effectiveness regardless of your genetic profile. Adequate sleep, stress management, regular exercise, and nutritious eating support your body’s healing processes. Share information about supplements, alternative therapies, and lifestyle factors with your healthcare team so they can consider the complete picture when personalizing your care.
The medical center and medical clinic department offer consultation services where you can discuss personalized options specific to your health concerns. Multidisciplinary teams collaborate to interpret test results and develop comprehensive treatment strategies.
Pro Tip: Keep open communication with your care team and consider second opinions if you’re uncertain about recommended tests or treatments, especially when significant costs or treatment changes are involved.
Explore personalized healthcare services at Globallmed
Understanding personalized healthcare concepts is just the beginning. Accessing these advanced approaches requires a healthcare partner equipped with modern diagnostic capabilities and experienced clinicians who can interpret complex results. Globallmed’s patient-centered approach integrates personalized healthcare principles into comprehensive treatment planning.
Our services span diagnostic testing, specialist consultations, and coordinated care across multiple disciplines. The medical clinic department provides access to physicians trained in genomic medicine who can order appropriate tests, interpret results, and modify treatment plans based on your unique profile. The multi therapy department offers integrated treatment options that address not just your medical condition but your overall wellbeing through coordinated therapeutic approaches.
Schedule a consultation to explore how personalized healthcare strategies might improve your treatment outcomes. Our team will assess your specific situation, explain relevant testing options, discuss costs transparently, and develop a tailored care plan that respects your preferences and goals.
What is personalized healthcare? Frequently asked questions
What types of conditions benefit most from personalized healthcare?
Cancer treatment shows the strongest evidence for personalized approaches, particularly lung, breast, colorectal, and melanoma cases with identifiable genetic mutations. Cardiovascular disease, psychiatric conditions, and chronic pain management also benefit from pharmacogenomic testing that predicts drug response. Rare genetic diseases often require personalized diagnosis and treatment strategies unavailable through standard protocols.
How much does personalized testing typically cost?
Basic pharmacogenomic panels cost $200 to $500, while comprehensive cancer tumor profiling ranges from $3,000 to $10,000. Whole genome sequencing can exceed $5,000 but prices continue declining as technology improves. Many factors affect final costs including test complexity, laboratory choice, and whether your insurance provides coverage.
Is personalized healthcare covered by insurance?
Coverage varies significantly by insurance plan, test type, and medical indication. Many insurers cover FDA-approved genetic tests for specific cancers or pharmacogenomic testing before starting certain medications. Pre-authorization is often required and coverage may be denied for tests considered experimental. Contact your insurance company before testing to understand your financial responsibility.
How do I talk to my doctor about personalized healthcare?
Start by asking whether genetic or biomarker testing would provide useful information for your condition. Request explanations of how test results might change your treatment plan and what evidence supports personalized approaches for your diagnosis. Bring questions about costs, turnaround time, and result interpretation to your appointment. If your doctor seems unfamiliar with personalized options, consider requesting a referral to a specialist or seeking a second opinion.
Are there risks or privacy concerns with genetic testing?
Genetic information reveals details about disease risks that may cause anxiety, and results sometimes have unclear significance. Privacy risks include potential data breaches exposing sensitive health information, though laws like GINA provide some protections against genetic discrimination. Your genetic data also reveals information about blood relatives who haven’t consented to testing. Discuss data storage, sharing policies, and privacy protections with your testing provider before proceeding.
